Trump Order Fast-Tracks Psychedelic Testing, Bypassing Regulators to Control Science from White House
To get a good sense of how policy gets made these days in Washington, look no further than Saturday’s signing event at the White House. The bottom line: Damn the clinical trials. Full speed ahead.
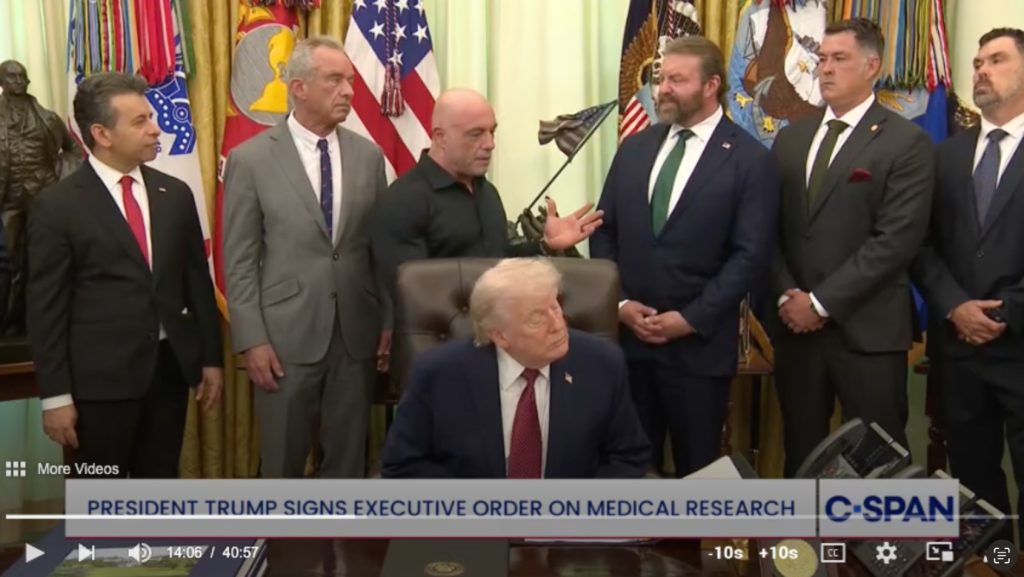
“Sounds great. Do you want FDA approval? Let’s do it.”
Those words, sent in a text message by the president of the United States to podcaster Joe Rogan, led to an extraordinary – can we still use that word in covering this administration? – executive order-signing and media event Saturday morning at the White House.
What a trip! In a flash, the decades-long long effort to end the overregulation of psychedelic drugs was seemingly over, and a new rush to race through – or past – the traditional regulatory processes of drug approvals began.
“Joe is an amazing guy, and he wrote me a little note about this, and I had it checked out,” Trump said at the signing.
Trump’s executive order directs the FDA and other federal agencies to speed up research on the therapeutic benefits of LSD, Ecstasy, psilocybin and other drugs and to ease restrictions that have limited the ability of scientists to study them.
It also directs Health and Human Services Secretary Robert F Kennedy Jr., who stood behind Trump at the signing, to provide $50 million “to match investment made by state governments” to accelerate research.
Texas is the only state to have initiated such research, which is focused on ibogaine, a powerful psychedelic made from a Central African plant that has attracted interest from researchers for its potential to treat substance abuse, including opioid use disorder.
It has been used in medicinal rituals for centuries in Gabon, and some studies have found that it rapidly cured people of addiction, but its use is also intense, lasts many hours and carries significant cardiac risks.
The order will also enable patients with serious mental illness to receive psychedelic drugs still in early clinical trials but not yet approved by the FDA as safe and effective.
“We’re taking this decision, this decisive step, to confront one of the most urgent public health challenges facing our nation, the mental health crisis,” Trump said.
At the event, Marcus Luttrell, the former Navy SEAL whose memoir about a deadly mission in Afghanistan was the basis of the film “Lone Survivor,” said psychedelics have helped him and other veterans struggling with mental health challenges.
“You’re going to save a lot of lives through it. It absolutely changed my life for the better,” he told Trump. “We had prayed for this day to come.”
Some observers were flabbergasted. “Stunned that the FDA Commissioner is in the back, acting as though this is normal,” tweeted Reshma Ramachandran, a physician and assistant professor at Yale School of Medicine. “(Why) even have an FDA to ensure that safety and efficacy is independently verified when you can just have a podcast host/influencer call up your boss and circumvent the process?”
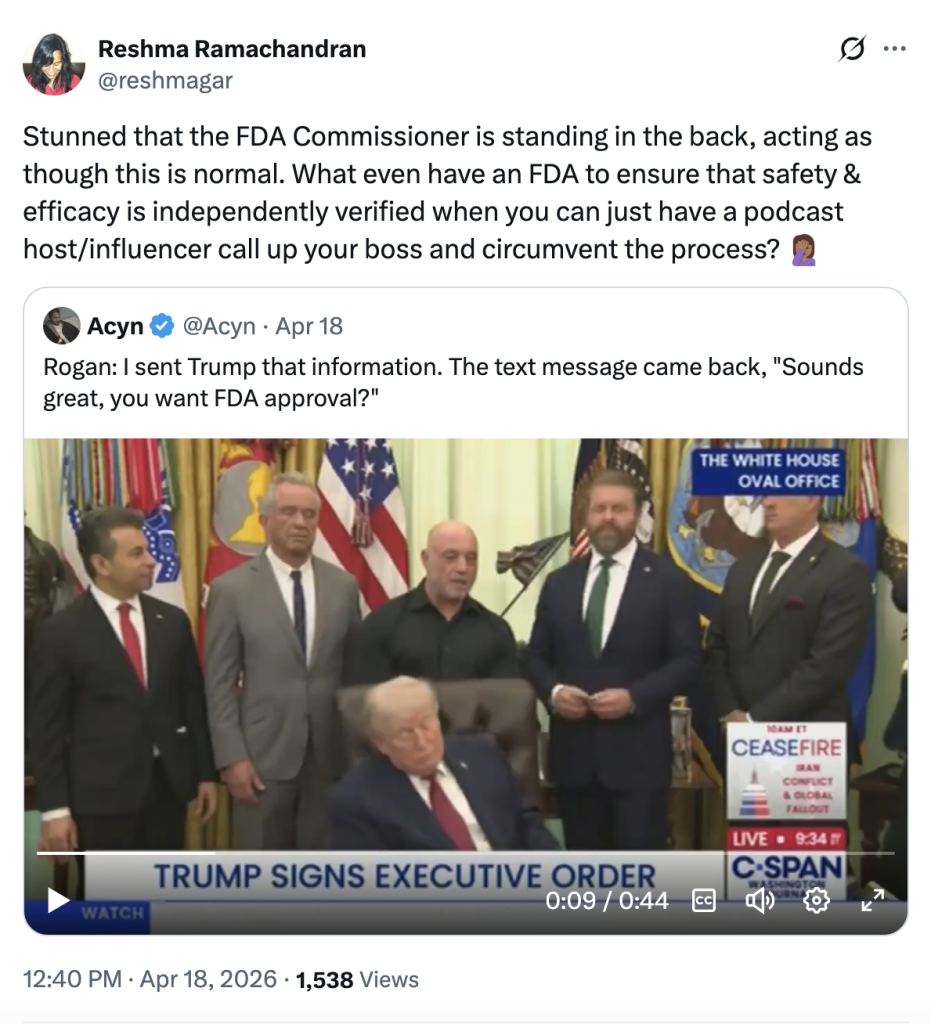
Trump said that after his text exchange with Rogan, he quickly reached out to his top health officials.
“I called Bobby, I called Oz, I called Marty and Jay,” the president said. “It was uniform support.”
Mehmet Oz, the physician and former TV personality who Trump tapped to head the Centers for Medicare and Medicaid Services, said the order was written in less than a week.
“The president just would not take no for an answer,” he said.
Gary Schwitzer, a longtime journalist and academic focused on media coverage of health and science, wrote on Substack that he was “stunned“ by the process.
“Yes, some psychedelics might be promising treatments for some hard-to-treat mental health conditions. But ‘might be’ must always be matched with ‘might not be.’ The potential benefits-to-harms ratio isn’t clear yet. And, yes, that’s why more research is needed.”
Then Schwitzer gave the last word to Trump’s own former surgeon general, Jerome Adams, who wrote on Twitter:
“The rollout was problematic. Announced in a WWE-style Oval Office presser complete with Joe Rogan, it included several mischaracterizations and exaggerations about the benefits of psychedelics and the legal FDA approval process. … Promising early data (e.g. Stanford MISTIC) must still go through rigorous, supervised trials. Public trust and patient safety require getting the facts and the regulatory process right.”
To read an extensive analysis of Trump’s executive order and where things go from here from Josh Hardman, an analyst who covers the world of psychedelics, go here.
The name “MindSite News” is used with the express permission of Mindsight Institute, an educational organization offering online learning and in-person workshops in the field of mental health and wellbeing. MindSite News and Mindsight Institute are separate, unaffiliated entities that are aligned in making science accessible and promoting mental health globally.





